

|
||
|
|
||
|
|
||
|
|
#11 | |
|
This is my second home
Rover 75 cdt club + Rover 2.5 KV6 Conni SE Join Date: May 2008
Location: Birmingham
Posts: 11,396
Thanks: 6,587
Thanked 2,262 Times in 1,729 Posts
|
Quote:
__________________
Great Barr, Birmingham. |
|
|
|

|
|
|
#12 |
|
This is my second home
15 years of 75s & ZTs now an XTrail Join Date: Nov 2006
Location: Danbury, Essex
Posts: 3,761
Thanks: 18
Thanked 88 Times in 56 Posts
|
Also more likely to leak, ?? Dave
__________________
ZT 190 X Power Grey The Only Colour is this Dark Silver!! When I was a kid I was scared of earwigs because I thought they actually lived in your ears. I was even more terrified of cockroaches! |
|
|

|
|
|
#13 |
|
This is my second home
Rover 75 cdt club + Rover 2.5 KV6 Conni SE Join Date: May 2008
Location: Birmingham
Posts: 11,396
Thanks: 6,587
Thanked 2,262 Times in 1,729 Posts
|
quite correct dave. antifreeze has a "searching" action. this means if there are any weak/thin spots in the rad/other areas, it will start to "weep" through these areas, usually shows up on the rad as a whitish/pinkish stain. will also show if clips are not tight, round the ends of water pipes.
__________________
Great Barr, Birmingham. |
|
|

|
|
|
#14 |
|
Gets stuck in
Rover 75 2.5 V6 Club Auto Join Date: Feb 2007
Location: Crewe
Posts: 898
Thanks: 0
Thanked 0 Times in 0 Posts
|
This is basically correct, neat antifreeze has a lower freezing point and a lower boiling point; that's why we have pressurised systems, as pressure raised the boiling point, but doesn't affect the freezing point, (or not much, anyway). Don't aero engines use neat antifreeze because of the very low temperatures at altitude ?
|
|
|

|
|
|
#15 |
|
This is my second home
Rover75 and Mreg Corsa. Join Date: Nov 2006
Location: Sumweer onat mote o'dust (Sagin)
Posts: 21,753
Thanks: 341
Thanked 3,660 Times in 2,924 Posts
|
This is a copy of a couple of my posts on t'other channel, where this topic is also discussed and it also highlighted several misconceptions about 'antifreeze'.
Here's a table showing how the freezing point of the Ethylene Glycol/water mixture reaches its minimum at 70% EG and then starts to rise again. The freezing point of pure EG is only -12C. The frost protection offered by the 50% OAT system is said to be down to around -45C. This is lower than the pure EG/water mix shown in the table and is probably the result of the various additives present in the formula. 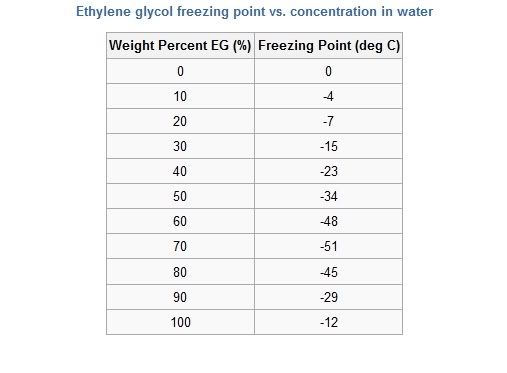 However, propylene glycol has a very much lower freezing point (-60C) and is widely used as a neat coolant for truck engines in America, Canada. Another misconception is that antifreeze greatly raises the boiling point of water, so it has less tendency to boil off in an overheat situation. It's also a misunderstanding that EG/water mixtures have a lower boiling point than water. They don't, as illustrated in the table below. The boiling point of typical coolant (50/50 EG/water) is only 107C, so not much higher than water alone. Here's another table showing how the boiling point varies with EG content. 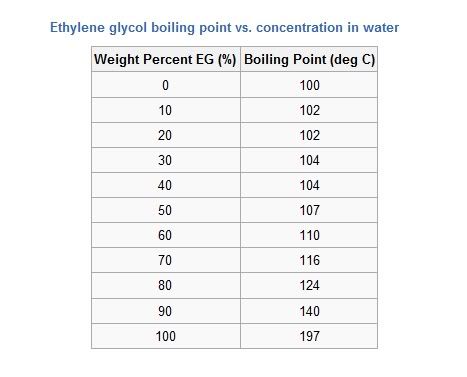 Here you can see that boiling point rises continously as the EG concentration is increased. It reaches the maximum at just under 200C for pure EG. The primary method of preventing boil up is to pressurise the system. This allows the coolant to get much hotter than 107C without boiling. Of course if you release the pressure cap when the temperature is at or above 107C, the whole thing will erupt instantaneously. TC Last edited by T-Cut; 20th January 2010 at 20:09.. |
|
|

|
 |
|
|